Latest Update
Latest Update - Thanks, Biopsies, and THE next study
- “Thank you guys for all your support – in all means possible- whether by posting, sharing or even contributing to the donations. With the generous contribution of people here and across channels we have closed the target for the next study.” “Thank you @Arcturus9for your generous contribution which will expedite our efforts- and help in reaching a significant milestone in treating hairloss.“ – Dr Taleb Barghouthi.
- While colleagues such as Dr Bloxham have an interest in an FUT procedure, Dr Barghouthi is considering a full FUE or injecting into the recipient area for his next study.
“As for future trials, I had one planned for the 24 th October, but I pushed him back because of the conference. I thought it would be important to follow him closely in the first few weeks and I didn’t want to be away in his initial recovery. We agreed to do it in November as he also has no travel commitments or difficulties with regular follow ups. We also have another patient who will be potentially having Verteporfin in November along with his procedure. I think the optimal dosing is not too far off. I am still keen on increasing the dose slightly- or at least in some areas. ” – Dr Barghouthi, November 1st 2023
- A 4mm biopsy was taken for both test and control areas in patient 1 and analysed by a pathologist . “Please be aware that the pathologist had very little insight as to what the test area and control areas are and that he only knew that it is an area of an extraction site. He has no insight about the trial whatsoever. I also randomly assigned which areas to take the biopsy from and was taken from the 0.32 mg injection and control sites.
Test: Sections show follicular hyperkeratosis, focal follicular plugging, No Lichenoid perifollicular lymphoid infiltrate, No dyskeratosis of the follicular epithelium, No perifollicular concentric fibrosis and minimal dermal fibrosis. 10 Hair follicles are seen. Three in anagen, 5 in catagen and 2 in telogen phase.
Control : Sections show follicular hyperkeratosis, focal follicular plugging, focal lichenoid perifollicular lymphoid cell infiltrate, No dyskeratosis of the follicular epithelium. Perifollicular concentric fibrosis in some hair follicles and focal dermal fibrosis. 5 hair follicles are seen, 1 in anagen, 2 in catagen and 2 in telogen phase.
I do think this is clearly a positive analysis on a cellular level. Of Course, we do have limitations in that we couldn’t biopsy all areas but I think it is a good result after all, especially the variation in dermal fibrosis and hair counts.” – Dr Taleb Barghouthi, March 19th 2023
- On April 1st 2023, Dr Barghouthi discussed his latest work with verteporfin in this interview:
Dr Barghouthi’s First Trial With Verteporfin
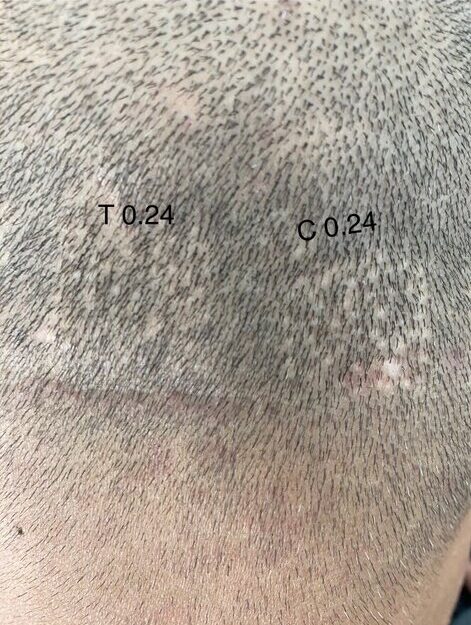
Dr. Barghouthi has tried using verteporfin on a “test” FUE. He set up 3 control and 3 test locations across the donor region, where the control was injected with PBS and the test locations were injected with verteporfin with a concentration of 2 mg/mL, which was the concentration used in pigs, with dosages of 0.24 mg/cm2, 0.32 mg/cm2, and 0.4 mg/cm2. These dosages were extrapolated from the mice study, as the dosages for the pig study were’nt known at the time of the trial. He’s seen the patient weekly for the first month, and around monthly since then. Below are the most recent pictures of the latest results after 16 months.
“I hope you’re keeping well. Apologies for keeping you waiting again. It was a busier than usual period in the last few weeks and I just landed in India for the annual hair conference.
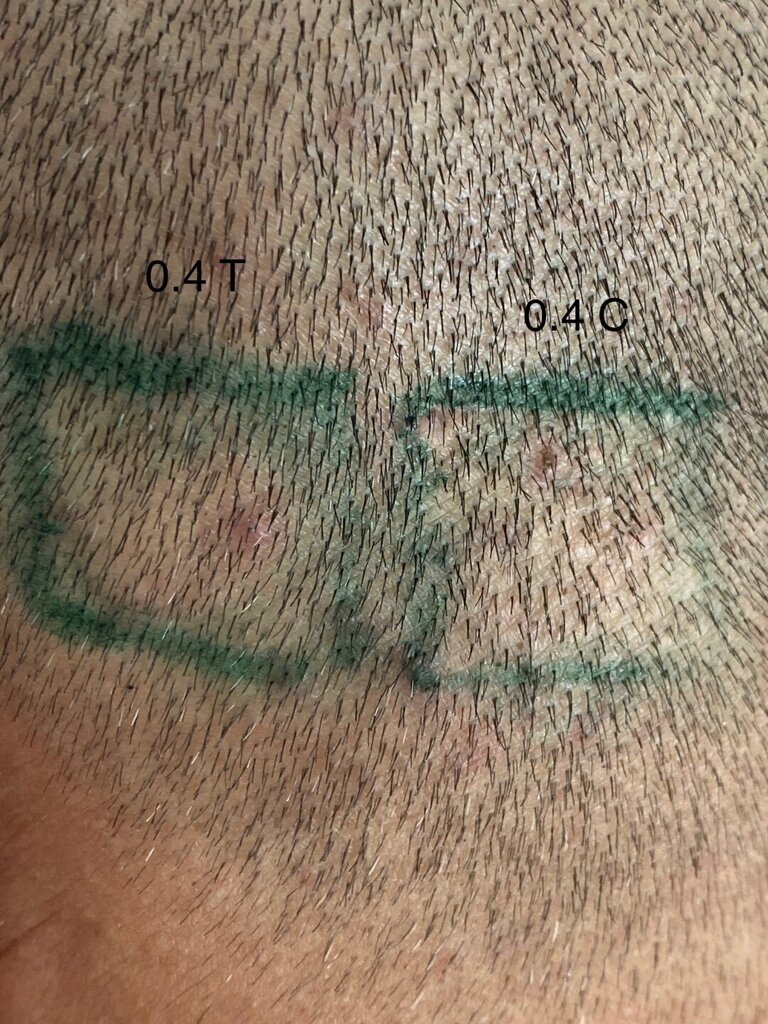
As mentioned in a previous post, I continue to see the consistent difference between injected and control sites. I think the 0.4 area has been showing the best overall progress- but I also think that we do notice changes in the other doses too. The zoomed out 0.4 area looks to me untouched- but that’s only my subjective judgement.
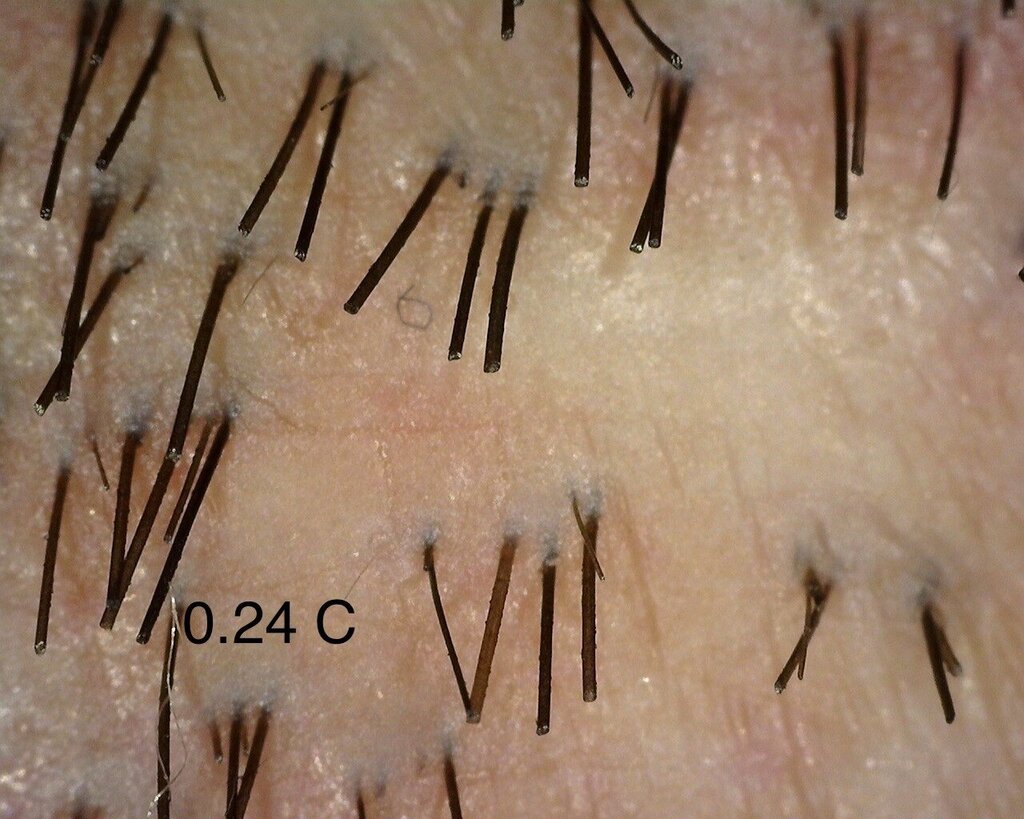
I was also happy to see the 0.24 mg area catching up to show better visual recovery and that might be due to the fact that this area had tumescent fluids injected in it, leading to vasoconstriction. This might be leading to a more localised effect of Verteporfin in the region.” – Dr Taleb Barghouthi, November 1st 2023
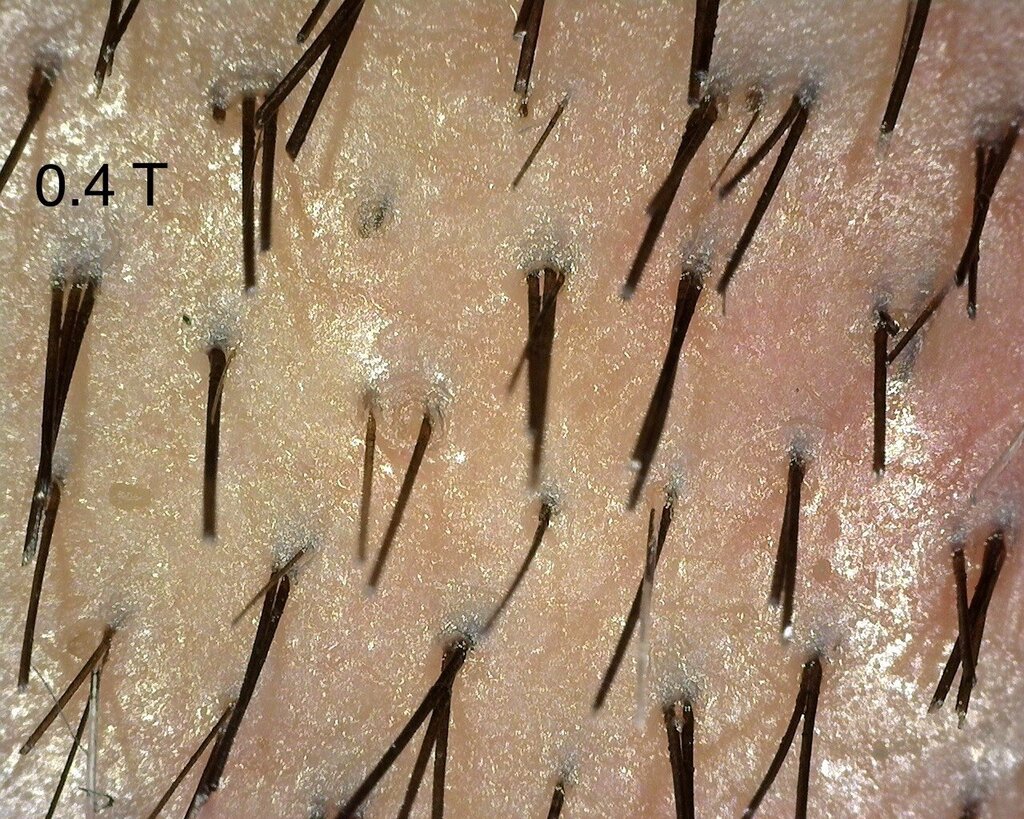
A close up of the 0.4 mg/cm² verteporfin treated area.
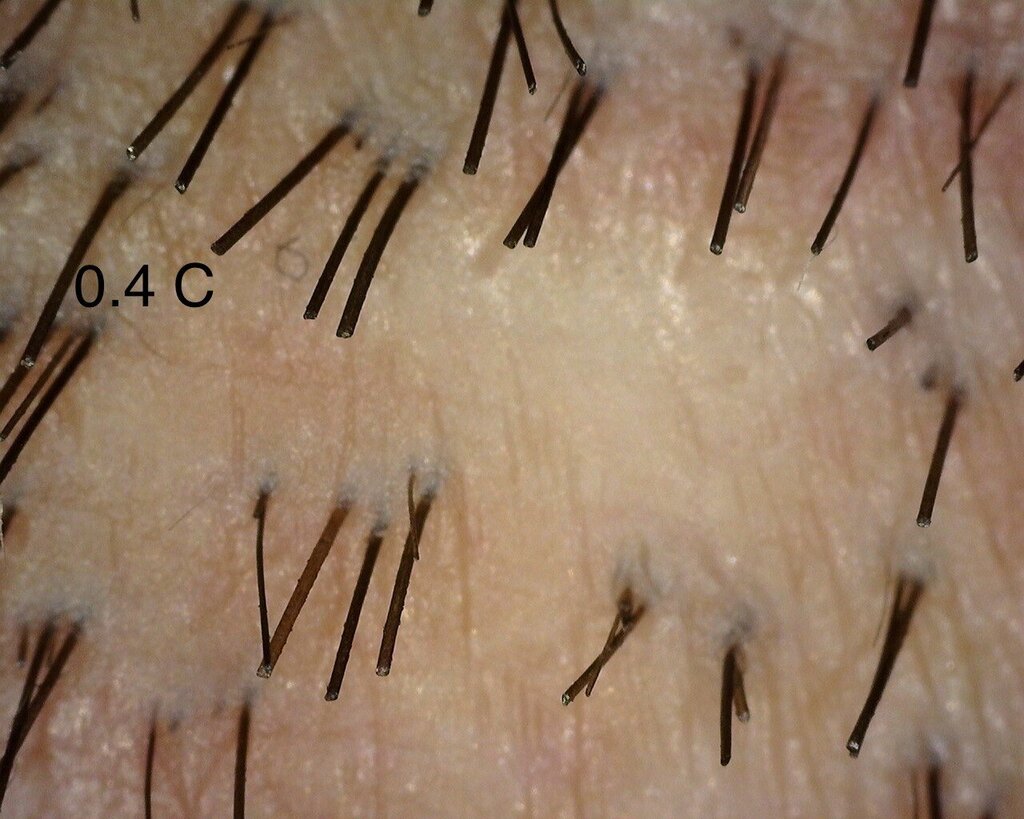
A close up of the 0.4 mg/cm² PBS treated control area.
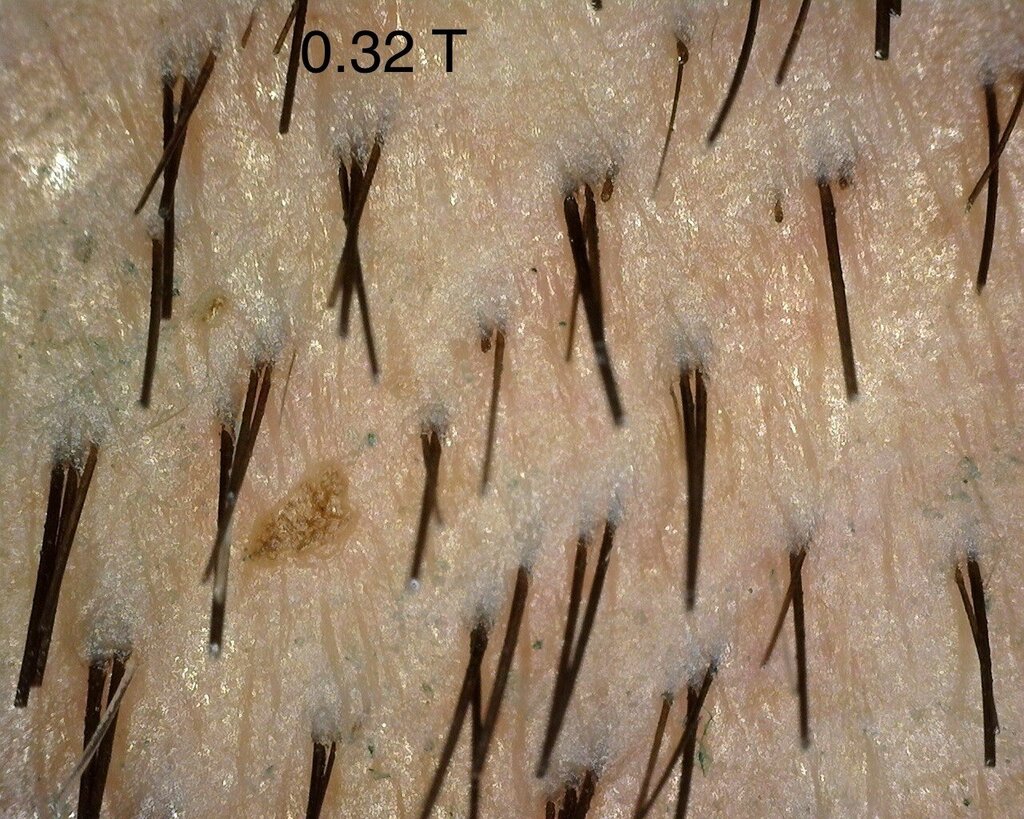
A close up of the 0.32 mg/cm² verteporfin treated area.

A close up of the 0.32 mg/cm² PBS treated control area.
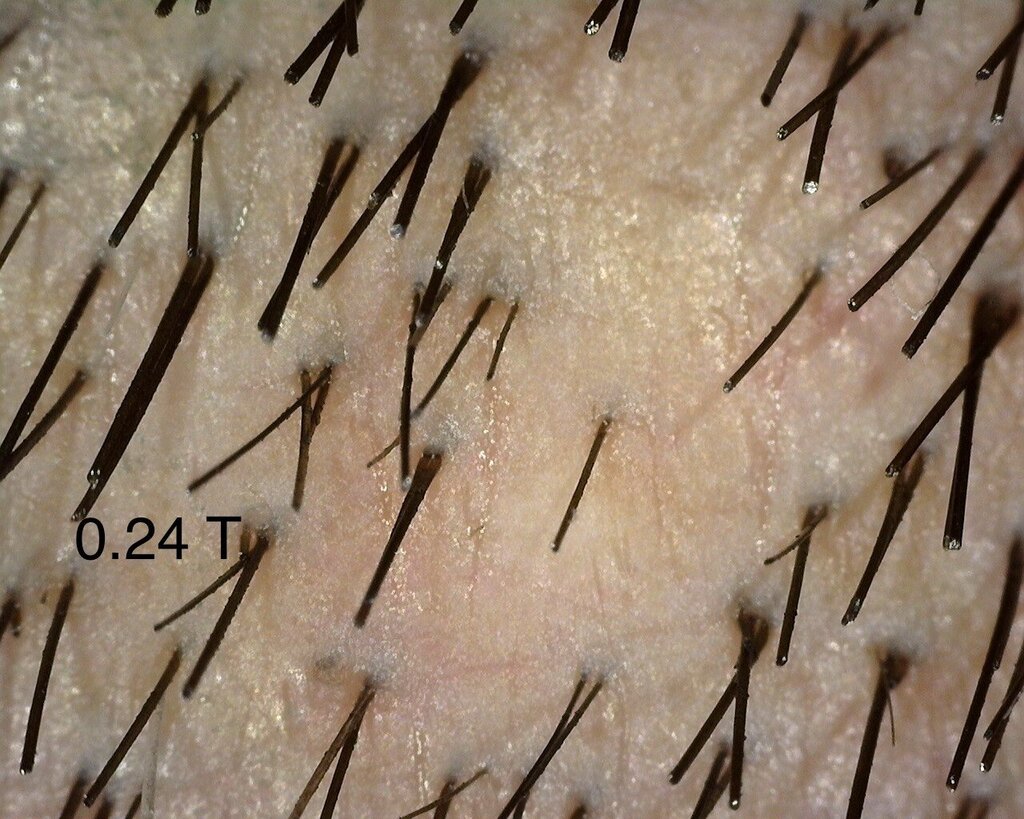
A close up of the 0.24 mg/cm² verteporfin treated area.